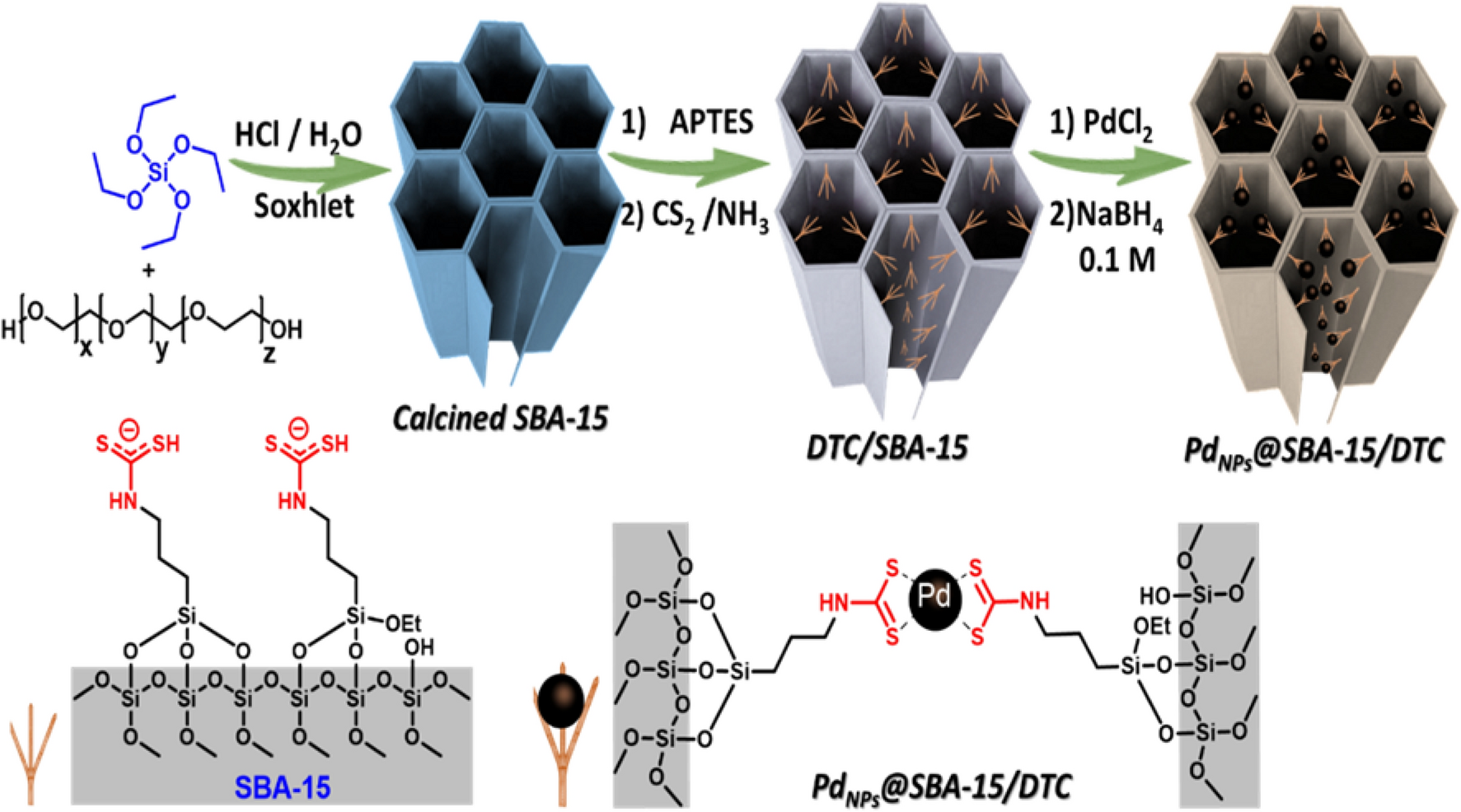
PDF) “Click” Polymer-Supported Palladium Nanoparticles as Highly Efficient Catalysts for Olefin Hydrogenation and Suzuki Coupling Reactions under Ambient Conditions | Didier Astruc - Academia.edu
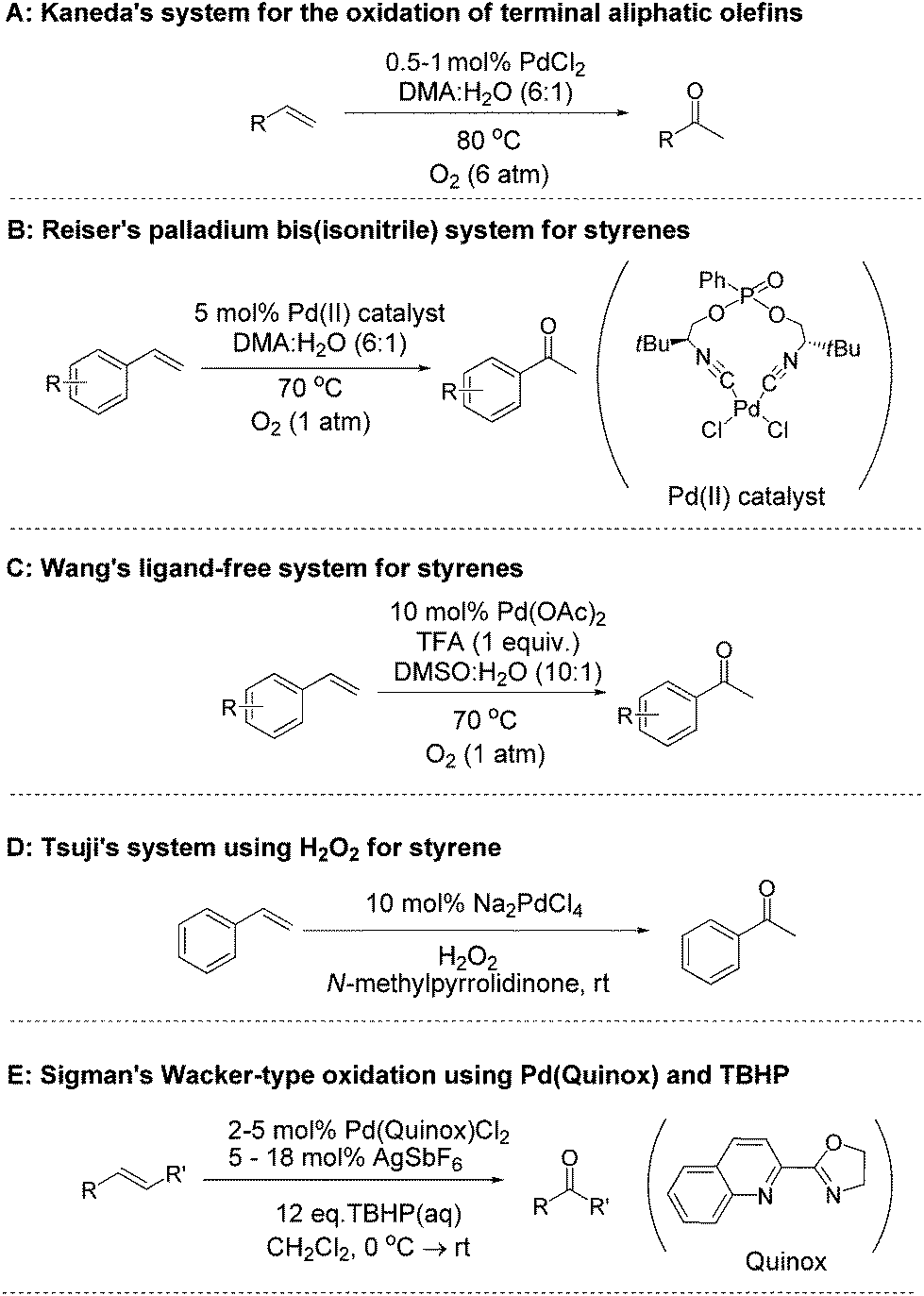
Cationic palladium( ii ) complexes as catalysts for the oxidation of terminal olefins to methyl ketones using hydrogen peroxide - Green Chemistry (RSC Publishing) DOI:10.1039/C4GC02465F

Combined Effect of Palladium Catalyst and the Alcohol to Promote the Uncommon Bis‐Alkoxycarbonylation of Allylic Substrates - Olivieri - 2022 - ChemCatChem - Wiley Online Library
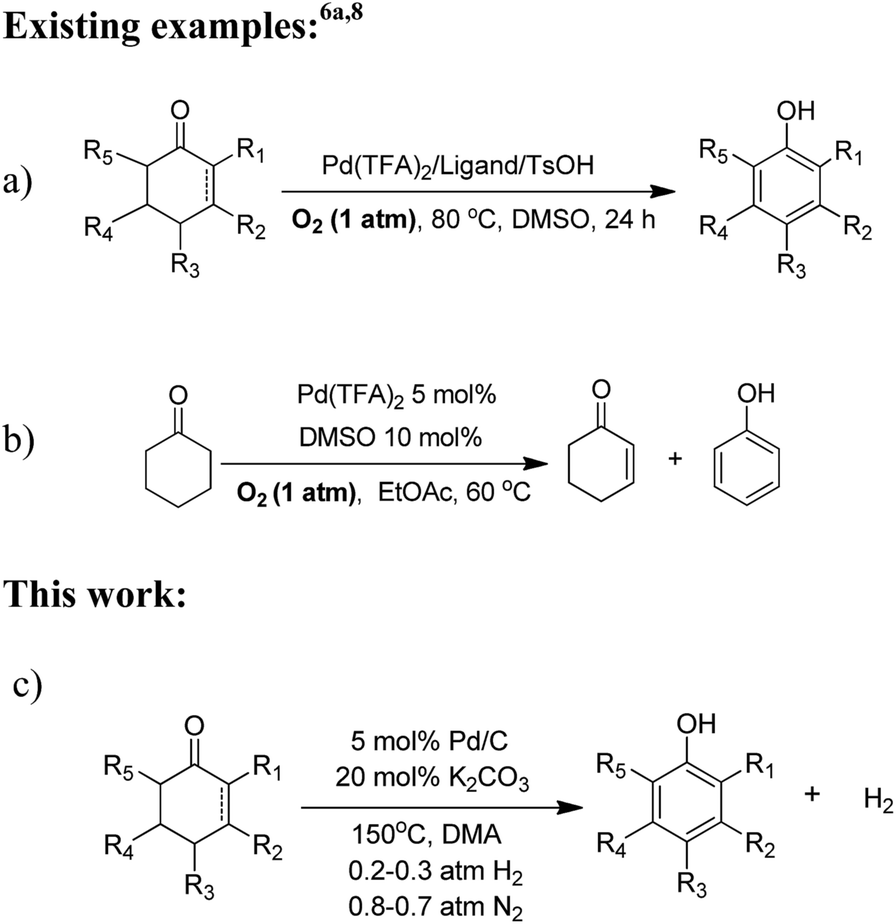
Reaction-activated palladium catalyst for dehydrogenation of substituted cyclohexanones to phenols and H 2 without oxidants and hydrogen acceptors - Chemical Science (RSC Publishing) DOI:10.1039/C5SC01044F
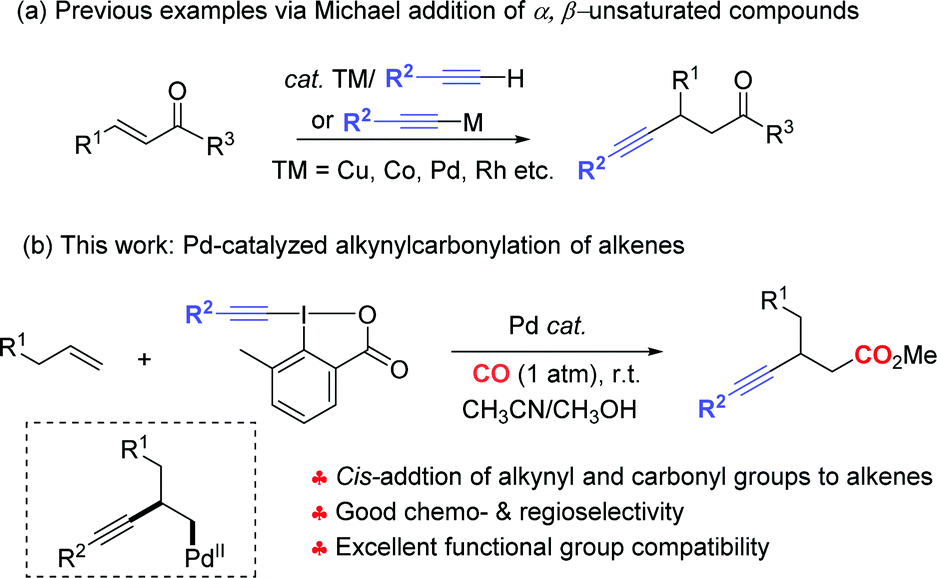
Palladium-catalyzed intermolecular alkynylcarbonylation of unactivated alkenes: easy access to β-alkynylcarboxylic esters - Chemical Communications (RSC Publishing) DOI:10.1039/D1CC07092D

High-Performance Nanostructured Palladium-Based Hydrogen Sensors—Current Limitations and Strategies for Their Mitigation | ACS Sensors

Recent Development in Palladium-Catalyzed Domino Reactions: Access to Materials and Biologically Important Carbo- and Heterocycles | Organometallics

Anchored Palladium Complex‐Generated Clusters on Zirconia: Efficiency in Reductive N‐Alkylation of Amines with Carbonyl Compounds under Hydrogen Atmosphere - Zhang - 2022 - Chemistry – An Asian Journal - Wiley Online Library

Non‐Chelate‐Assisted Palladium‐Catalyzed Aerobic Oxidative Heck Reaction of Fluorobenzenes and Other Arenes: When Does the C−H Activation Need Help? - ScienceDirect
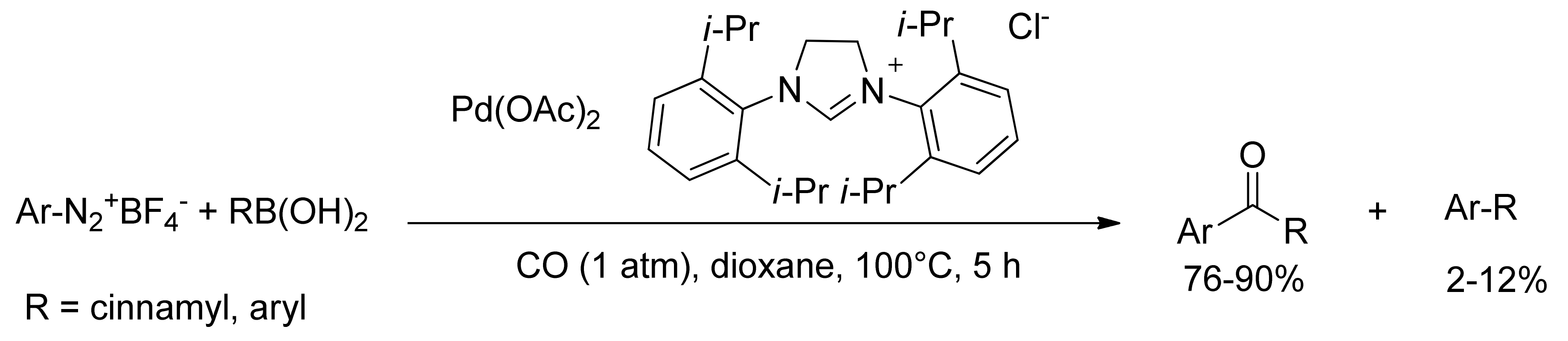
Molecules | Free Full-Text | Suzuki-Miyaura Cross-Coupling in Acylation Reactions, Scope and Recent Developments | HTML

Allylic C-H acetoxylation with a 4,5-diazafluorenone-ligated palladium catalyst: a ligand-based strategy to achieve aerobic catalytic turnover. - Abstract - Europe PMC
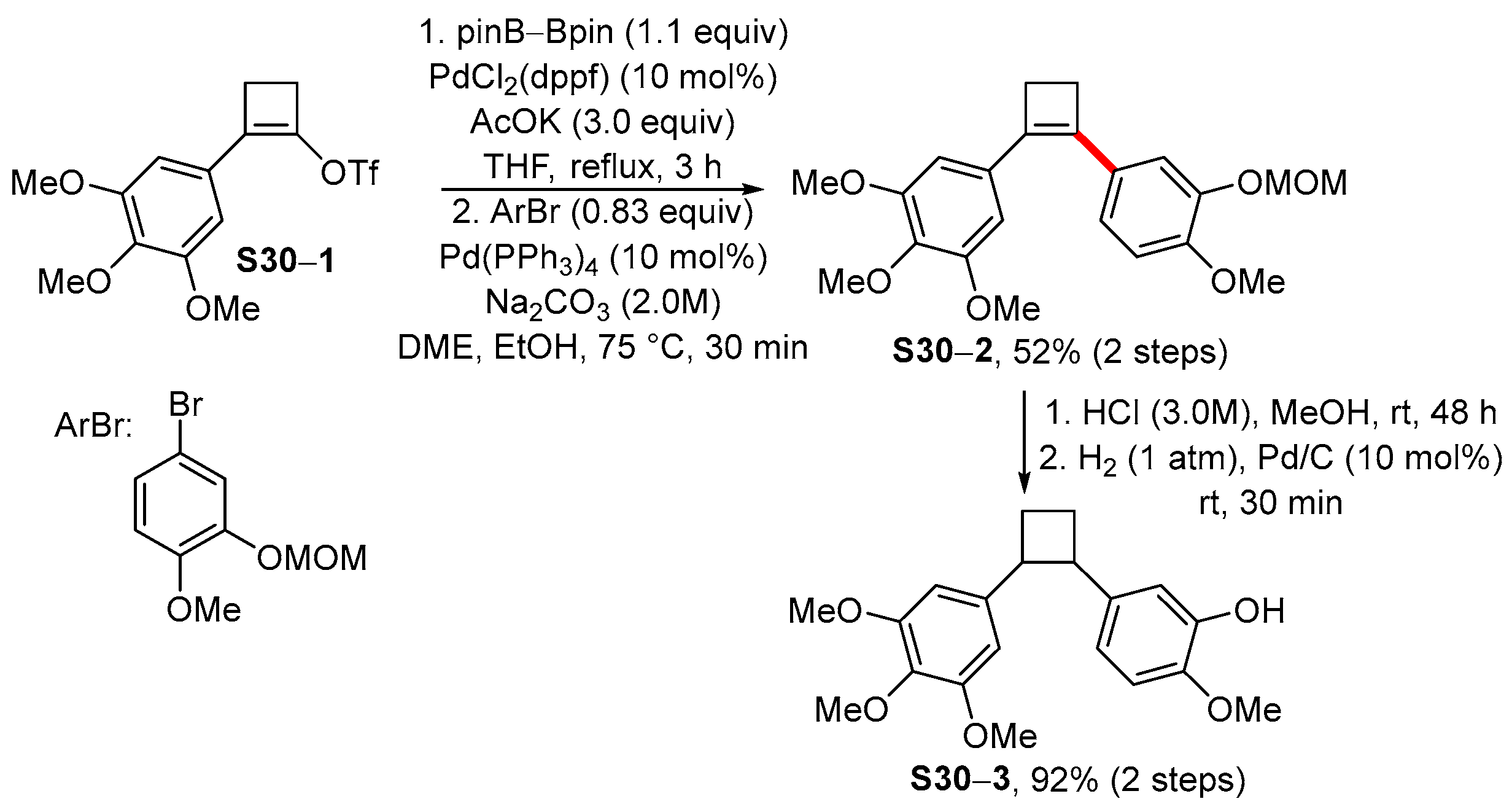
Organics | Free Full-Text | Palladium-Catalyzed Cross-Coupling Reactions of Borylated Alkenes for the Stereoselective Synthesis of Tetrasubstituted Double Bond | HTML

Palladium‐Catalyzed Alkoxy‐ and Aminocarbonylation of α‐Halomethyl Oxime Ethers: Synthesis of 1,3‐Alkoxyimino Esters and 1,3‐Alkoxyimino Amides - Noverges - 2015 - Advanced Synthesis & Catalysis - Wiley Online Library