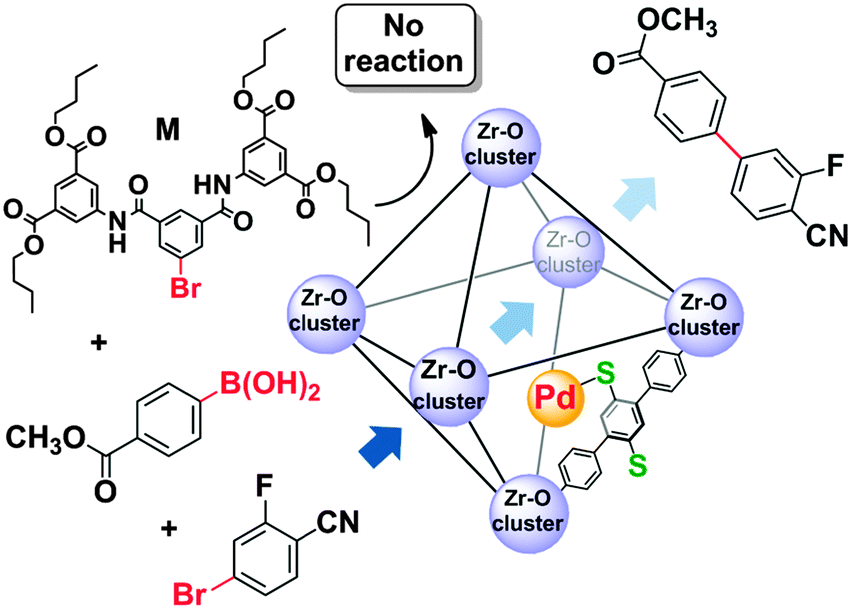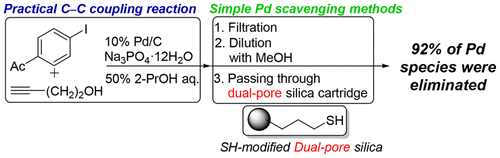
Application of Thiol-Modified Dual-Pore Silica Beads as a Practical Scavenger of Leached Palladium Catalyst in C–C Coupling Reactions - Org. Process Res. Dev. - X-MOL
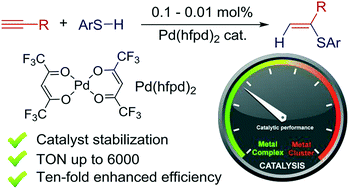
Ten-fold boost of catalytic performance in thiol–yne click reaction enabled by a palladium diketonate complex with a hexafluoroacetylacetonate ligand - Catalysis Science & Technology (RSC Publishing)

Effects of two different thiol ligands to form a mixed monolayer ligand... | Download Scientific Diagram

Synthesis, characterization, structural studies, DNA interaction, and cytotoxic studies of palladium(II) mixed-ligand complexes containing 2,2′-bipyridine, 5,6-dimethyl-1,10-phenanthroline and tetrazole-5-thiol ligands - ScienceDirect
Synthesis of Alkanethiolate-Capped Metal Nanoparticles Using Alkyl Thiosulfate Ligand Precursors: A Method to Generate Promising
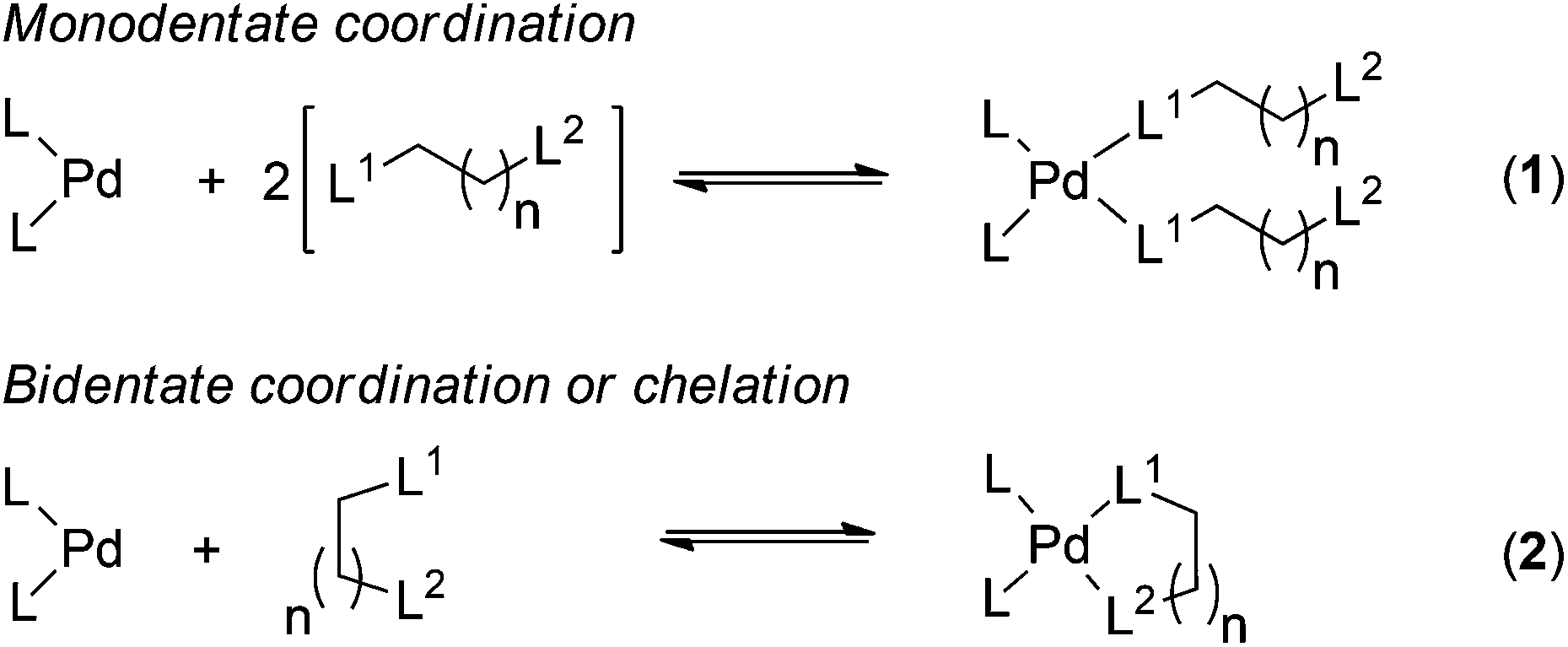
Towards a quantitative understanding of palladium metal scavenger performance: an electronic structure calculation approach - Dalton Transactions (RSC Publishing) DOI:10.1039/C3DT52282B
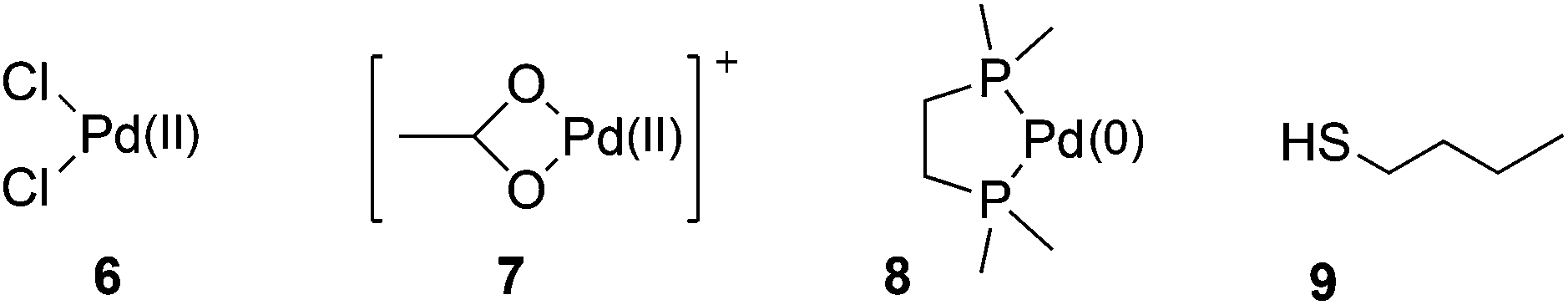
Towards a quantitative understanding of palladium metal scavenger performance: an electronic structure calculation approach - Dalton Transactions (RSC Publishing) DOI:10.1039/C3DT52282B

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect

Pd/BIPHEPHOS is an Efficient Catalyst for the Pd‐Catalyzed S‐Allylation of Thiols with High n‐Selectivity - Schlatzer - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
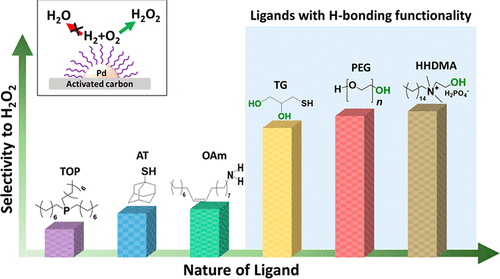
Tunable Catalytic Performance of Palladium Nanoparticles for H2O2 Direct Synthesis via Surface-Bound Ligands - ACS Catal. - X-MOL

Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation | Science

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -

Alkyne Hydrothiolation Catalyzed by a Dichlorobis(aminophosphine) Complex of Palladium: Selective Formation of cis‐Configured Vinyl Thioethers - Gerber - 2012 - Chemistry – A European Journal - Wiley Online Library
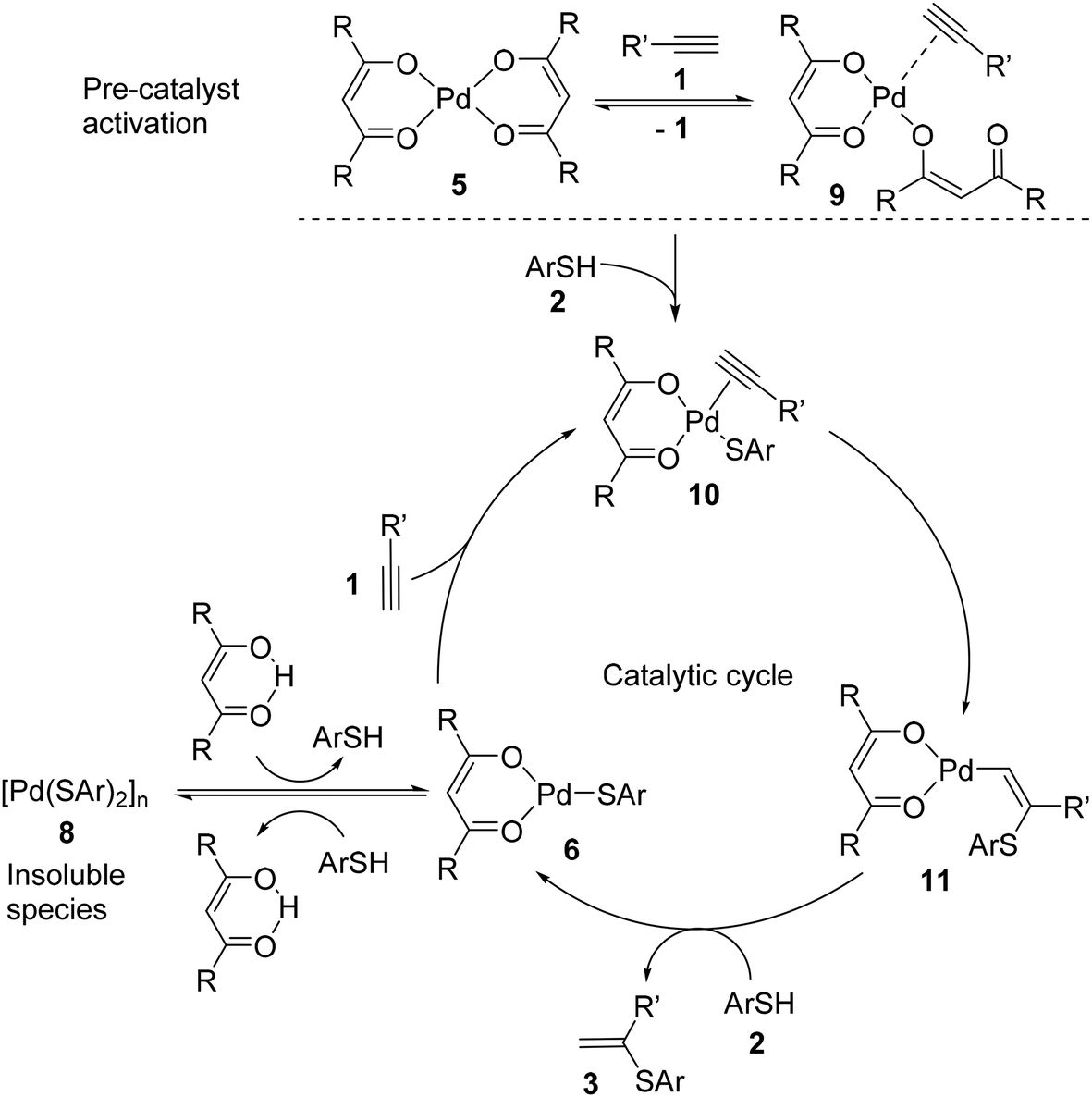
Ten-fold boost of catalytic performance in thiol–yne click reaction enabled by a palladium diketonate complex with a hexafluoroacetylacetonate ligand ... - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C8CY00173A

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect
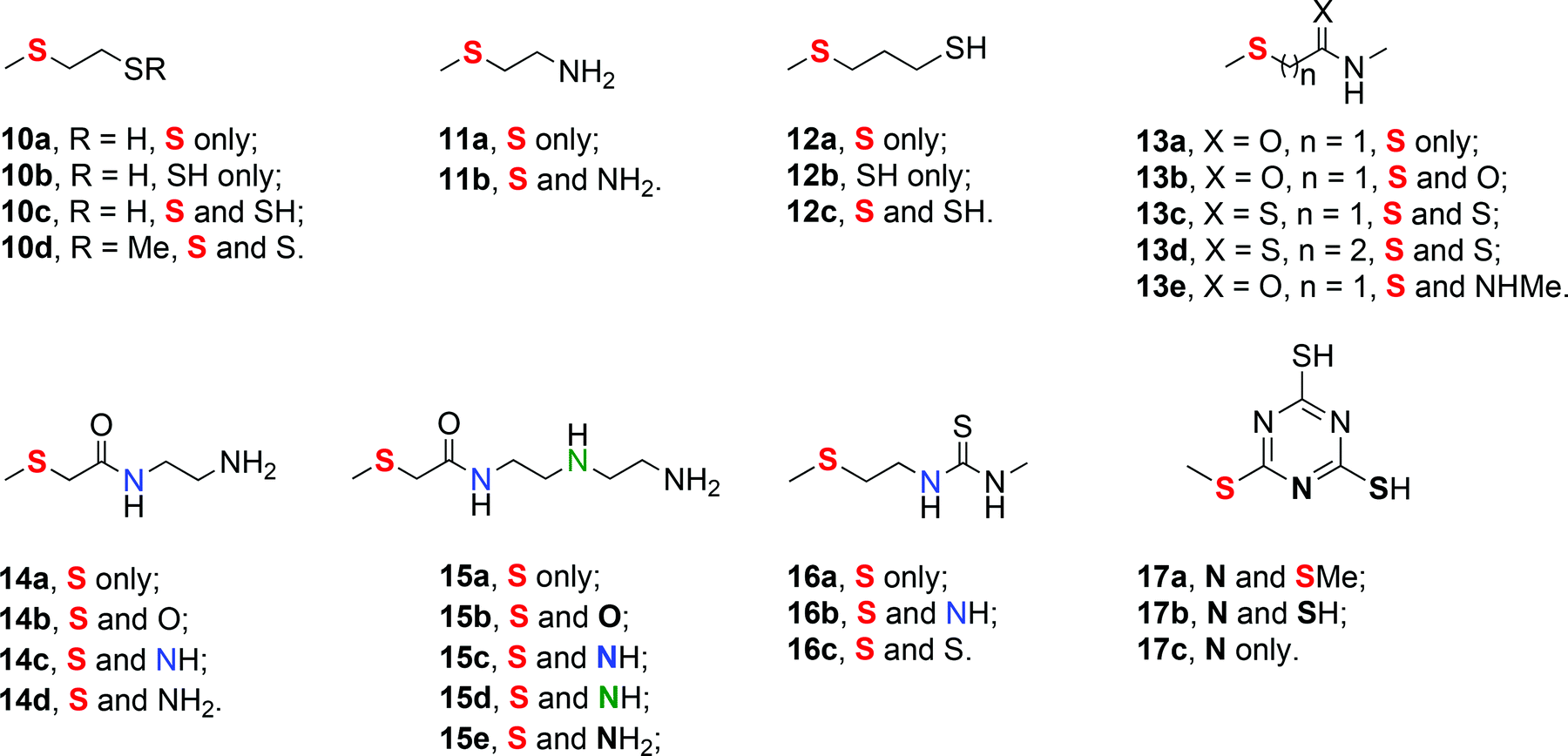
Towards a quantitative understanding of palladium metal scavenger performance: an electronic structure calculation approach - Dalton Transactions (RSC Publishing) DOI:10.1039/C3DT52282B
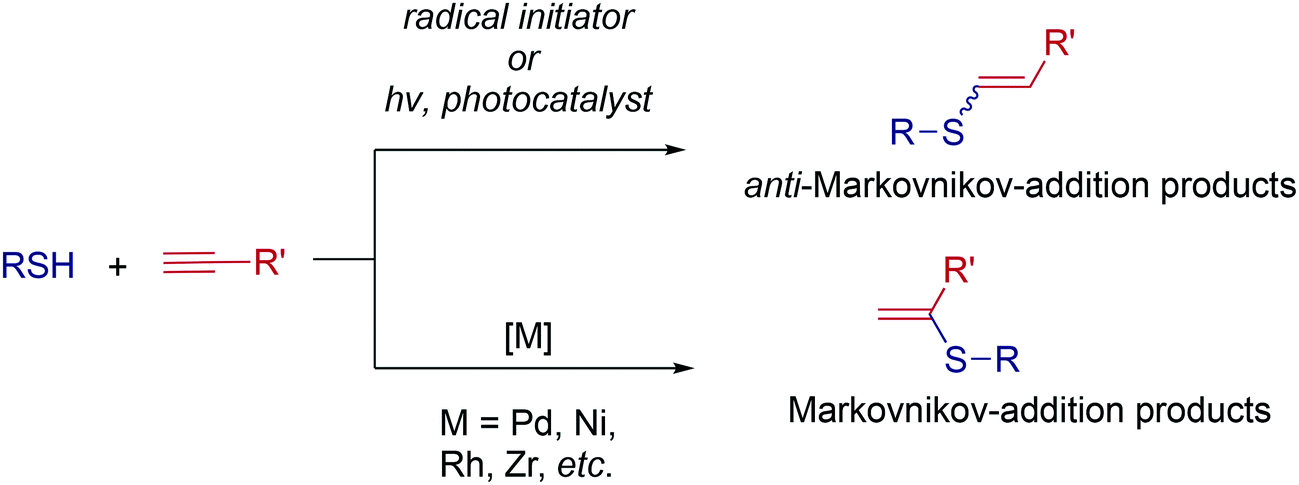
Ten-fold boost of catalytic performance in thiol–yne click reaction enabled by a palladium diketonate complex with a hexafluoroacetylacetonate ligand - Catalysis Science & Technology (RSC Publishing)
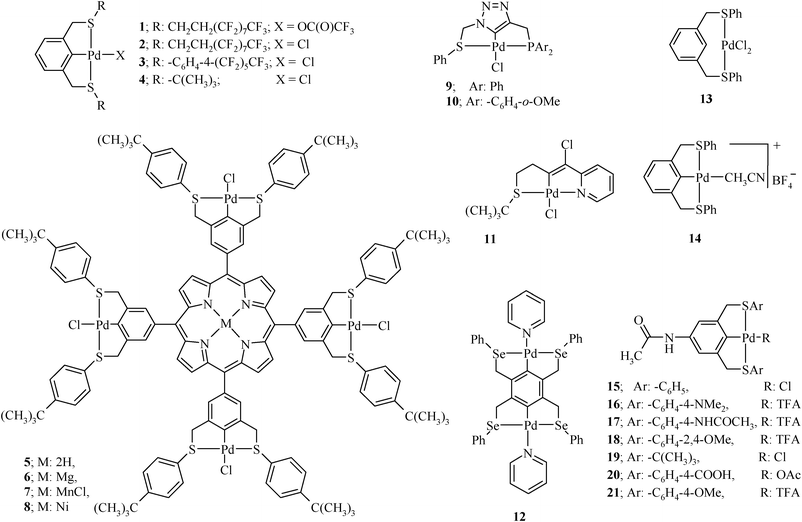
Organochalcogen ligands and their palladium( ii ) complexes: Synthesis to catalytic activity for Heck coupling - RSC Advances (RSC Publishing) DOI:10.1039/C2RA20508D
Synthesis of Alkanethiolate-Capped Metal Nanoparticles Using Alkyl Thiosulfate Ligand Precursors: A Method to Generate Promising