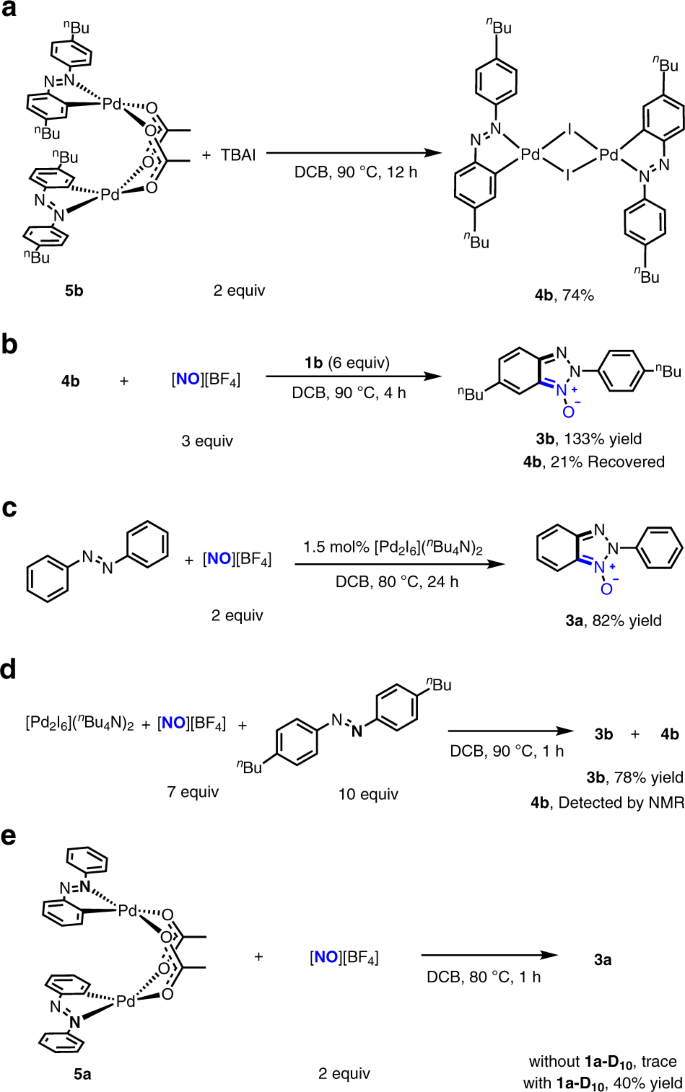
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Switching the Reactivity of Palladium Diimines with “Ancillary” Ligand to Select between Olefin Polymerization, Branching Regulation, or Olefin Isomerization - Jones - - Angewandte Chemie - Wiley Online Library

Palladium complexes of C/Se and N/E (E = Se, Te) ligands, 2-(3,5-dimethylpyrazol-1-yl)phenyl substituted chalcogeno-ethers: Functional group effect on structure and catalytic activity - ScienceDirect

Developing ligands for palladium(II)-catalyzed C-H functionalization: intimate dialogue between ligand and substrate. - Abstract - Europe PMC

The mechanism of palladium(II)-mediated C–H cleavage with mono-N-protected amino acid (MPAA) ligands: origins of rate acceleration in: Pure and Applied Chemistry Volume 88 Issue 1-2 (2016)
Cationic Two-Coordinate Complexes of Pd(I) and Pt(I) Have Longer Metal- Ligand Bonds Than Their Neutral Counterparts

Molecular ligand modulation of palladium nanocatalysts for highly efficient and robust heterogeneous oxidation of cyclohexenone to phenol | Science Advances

Solvent effects on ligand exchange between two different pd(II) organometallic complexes - ScienceDirect

The mechanism of palladium(II)-mediated C–H cleavage with mono-N-protected amino acid (MPAA) ligands: origins of rate acceleration in: Pure and Applied Chemistry Volume 88 Issue 1-2 (2016)
Neutral, cationic and anionic organonickel and -palladium complexes supported by iminophosphine/phosphinoenaminato ligands - Dalton Transactions (RSC Publishing)

Ligand characteristics and in situ generation of Pd active species towards CC coupling using series of 2-(1H-imidazol-2-yl)phenols - ScienceDirect

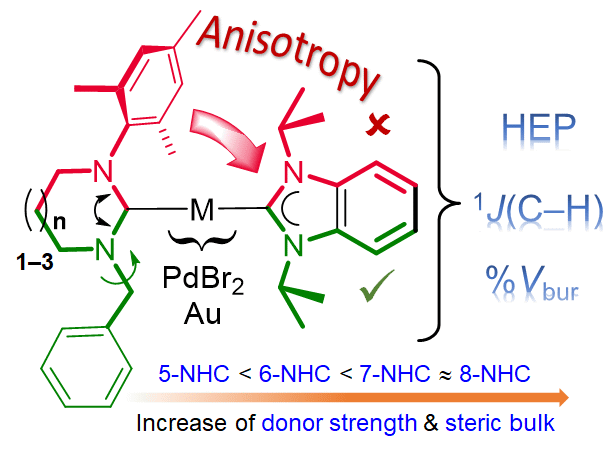
Platinum(II), palladium(II) and gold(I) benzimidazolin-2-ylidene as potential probes for determination of N-heterocyclic carbene donor strengths and steric bulks by DFT calculations - J. Chem. Sci. - X-MOL

Developing backbone-modified Mor-DalPhos ligand variants for use in palladium-catalyzed C–N and C–C cross-coupling
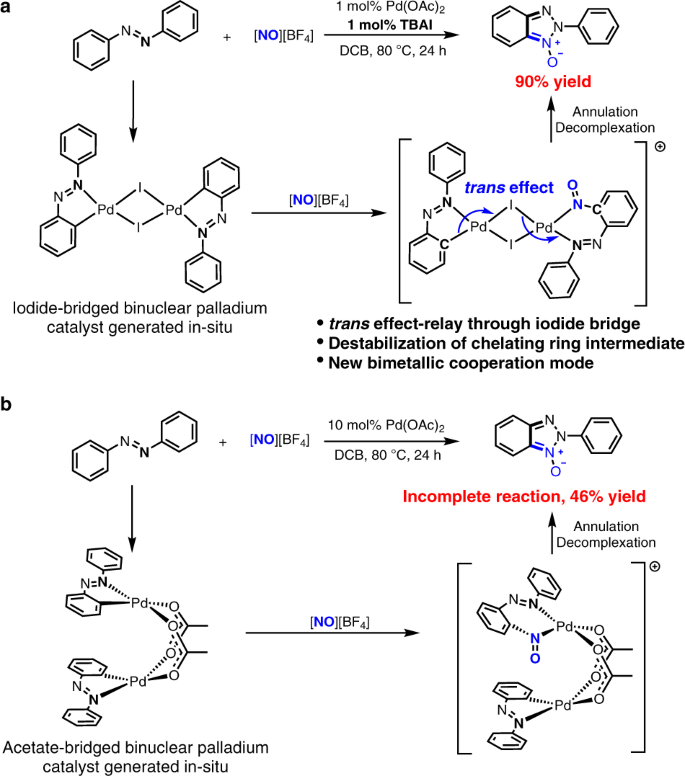
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Developing ligands for palladium(II)-catalyzed C-H functionalization: intimate dialogue between ligand and substrate. - Abstract - Europe PMC
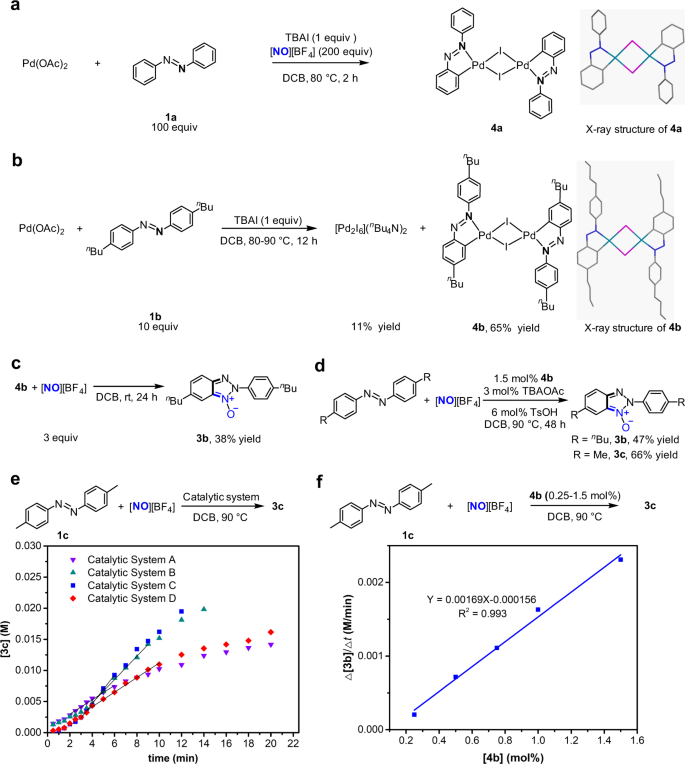
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
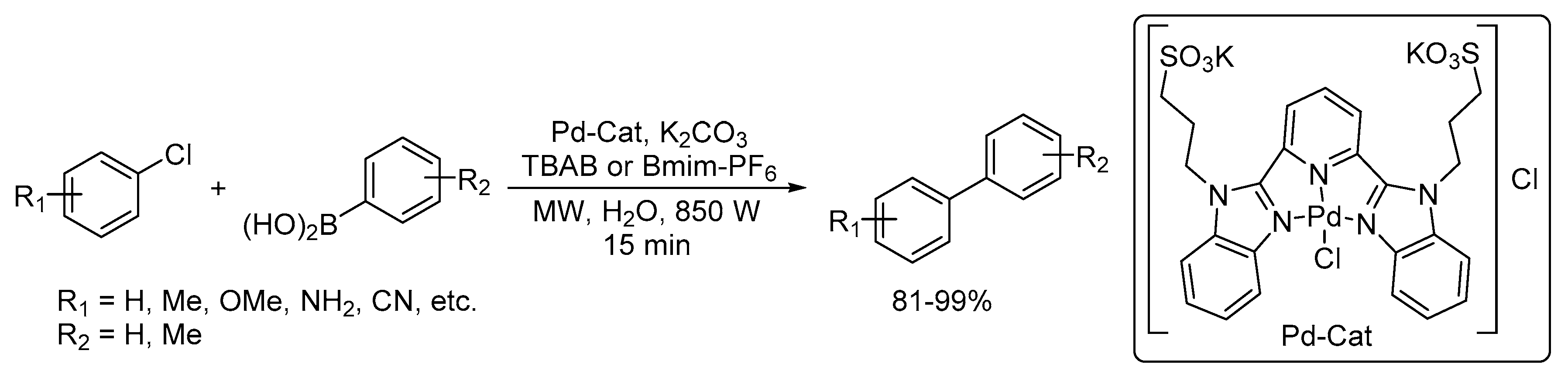
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Cationic Two-Coordinate Complexes of Pd(I) and Pt(I) Have Longer Metal- Ligand Bonds Than Their Neutral Counterparts - ScienceDirect

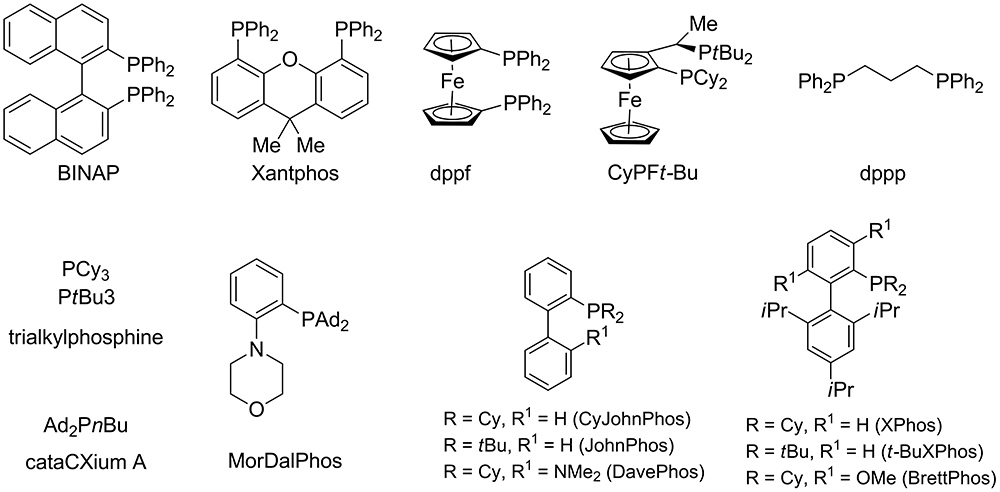

![Palladium Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | TCI EUROPE N.V. Palladium Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | TCI EUROPE N.V.](https://www.tcichemicals.com/assets/cms-images/03.gif)

