Strategies To Control Product Characteristics in Simultaneous Crystallization of NaCl and KCl from Aqueous Solution: Seeding with KCl | Crystal Growth & Design

Question Video: Determining the Mass of the Potassium Chloride Analyte When 2g of Silver Chloride Precipitates | Nagwa

A particularly simple NH4Cl-based method for the dissolution of UO2 and rare earth oxides in LiCl-KCl melt under air atmosphere - ScienceDirect

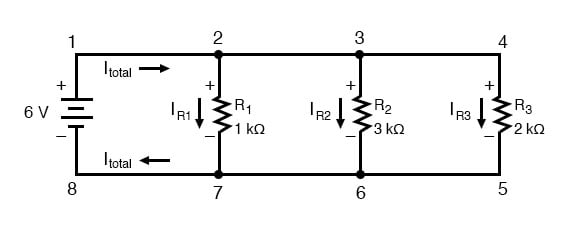
SOLVED: Use the following information to calculate the heat of sublimation for potassium (K): Heat of formation for KCl(s) = -437 kJ/mol Electron affinity for Cl = -349 kJ/mol Ionization energy for

Atomic insights into flotation separation of KCl and NaCl from a new viewpoint of hydration layer: A molecular dynamic study - ScienceDirect
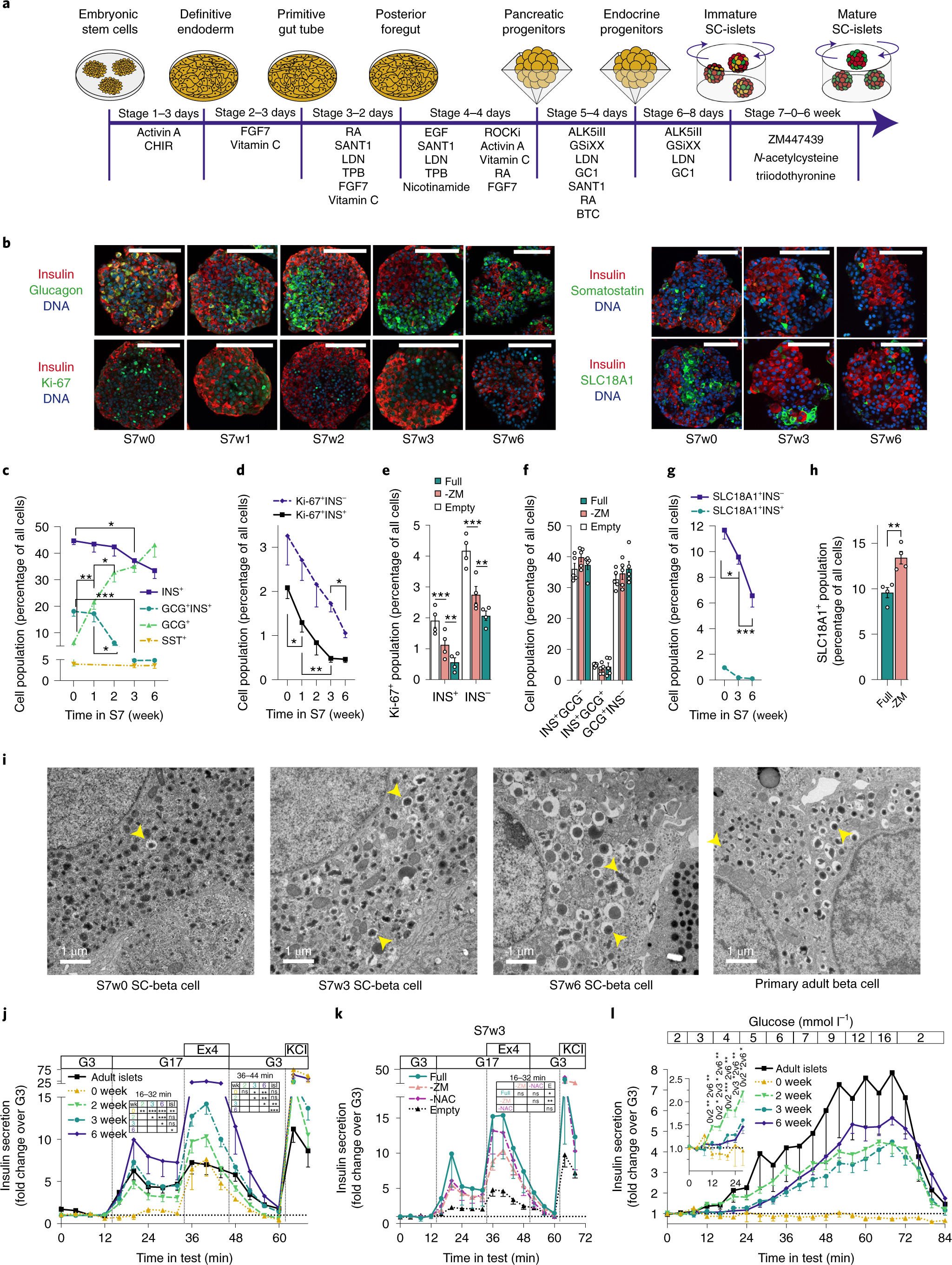
Functional, metabolic and transcriptional maturation of human pancreatic islets derived from stem cells | Nature Biotechnology

NaCl and KCl mediate log increase in AAV vector particles and infectious titers in a specific/timely manner with the HSV platform - ScienceDirect
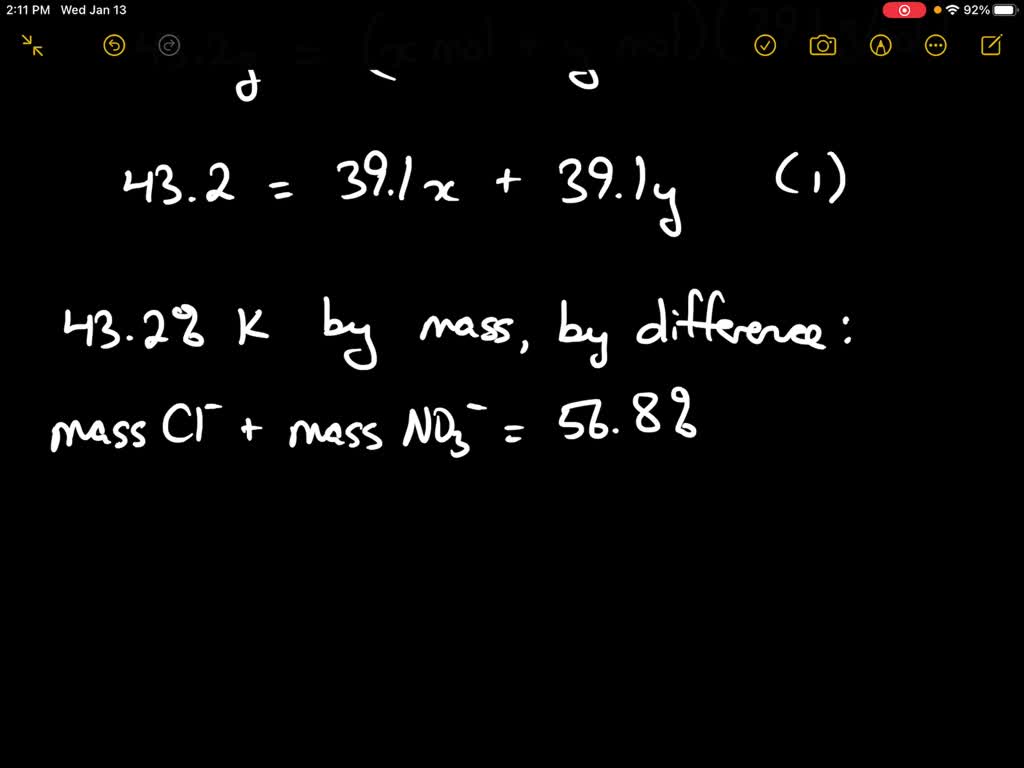
SOLVED: El peso molecular del KCl (Cloruro de potasio)es: * 74,54 g/mol 35,45 g/mol 73, 54 g/mol 46,45 g/mol